Connecting the region's cancer centers to global clinical programs.
Partner with Us
Atla turns clinical data inside Latin American cancer centers into queryable research assets.
Longitudinal, de-identified patient data from oncology populations largely absent in existing research databases.
Genetic backgrounds, disease presentations, and treatment patterns absent from US and European datasets.
Detailed treatment trajectories from diagnosis through follow-up, including lines of therapy and outcomes.
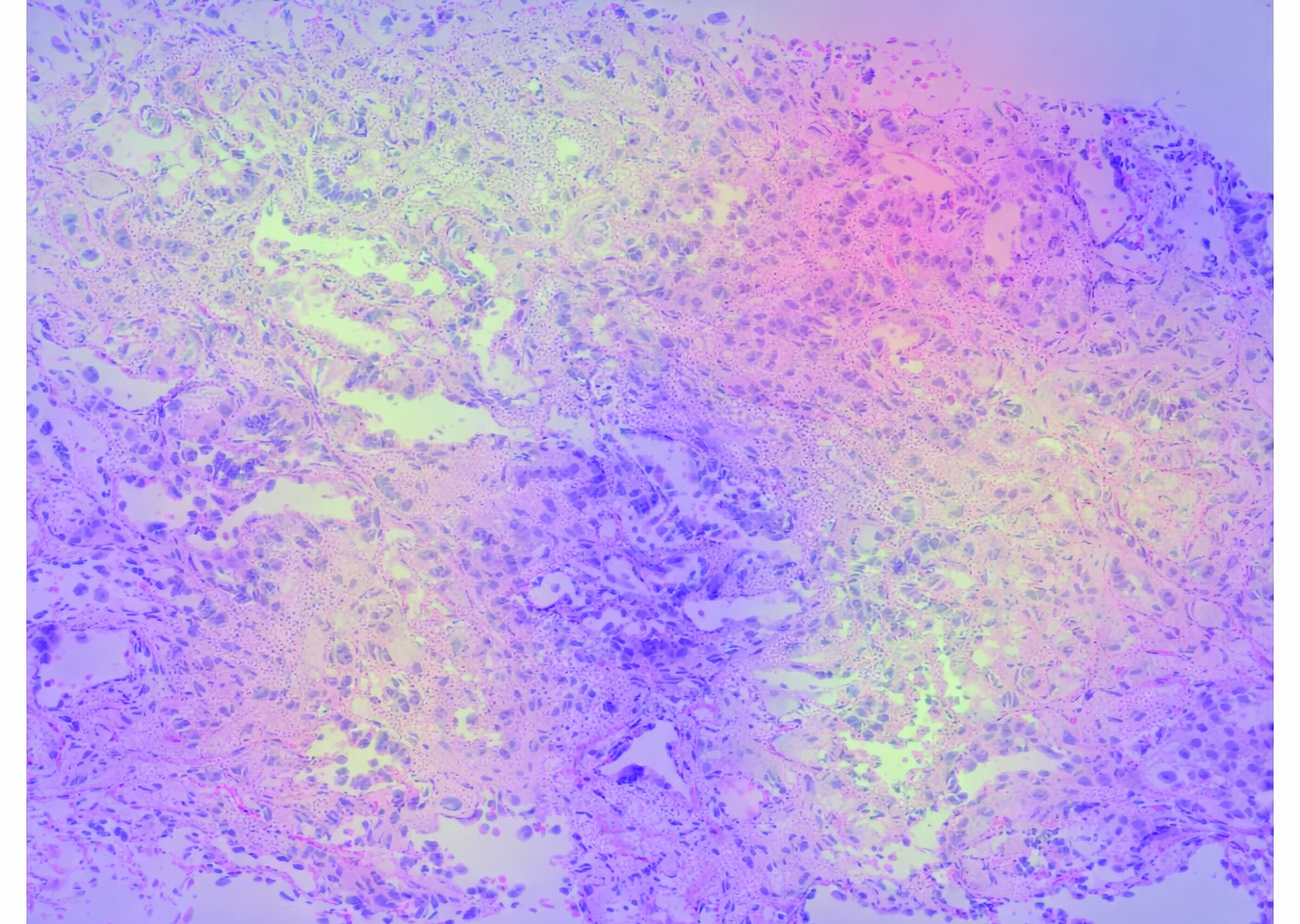
Pathology slides with model predictions linked to clinical outcomes. Designed for external validation or fine-tuning on underrepresented populations.
627 eligible patients identified across 4 institutions. Median enrollment projection: 14 weeks. All sites are IRB-compliant with structured PD-L1 data 1.
Identify eligible patient populations across the network before committing to a site or a timeline.
Test inclusion/exclusion criteria against real institutional data before finalizing your study design.
Rank institutions by eligible patient volume, data quality, and regulatory readiness.
Subtype distribution, treatment lines, and demographic composition across the network.
Institutions with structured data pipelines, regulatory infrastructure, and pre-identified patient populations matching your protocol criteria.
Sites identify eligible candidates automatically against your protocol criteria, automating initial screening.
Deploy and validate AI models against diverse populations before regulatory submission. Run inference inside the network, not outside it.
Data mapped to SDTM, ADaM, and OMOP standards. Submission-ready from the start.
Integrated directly into institutional workflows.
Cancer patients across Latin America represent genetic backgrounds, disease presentations, and treatment patterns that US and European datasets do not capture.
Detailed histories from diagnosis through follow-up, including lines of therapy, response, progression, and long-term outcomes.
Samples linked to the complete clinical history of the patient they came from. Available for validation, follow-up analysis, and biological grounding.
Clinical data preserved in its original clinical context and mapped to OMOP, FHIR, SDTM, and ADaM for international research compatibility.
Run pathology, imaging, and clinical AI models inside the network. Validate against populations absent from your training data, or deploy licensed models into institutional workflows.
Every participating institution chooses which studies to join and which partners to engage. Access is permissioned, queries are logged, and consent is explicit.